The online platform facilitating the access to clinical trials in Romania has been launched
Patients in Romania, their relatives, the medical community and the general public can access information about ongoing clinical trials in Romania, through a new online platform, in Romanian, which is connected to the European Union database.
The National Agency for Medicines and Medical Devices of Romania (NAMMDR) together with the Health Innovation Hub have developed and launched the online platform facilitating the access to clinical trials in Romania, available at https://studiiclinice.anm.ro/.
download the press release… ![]()
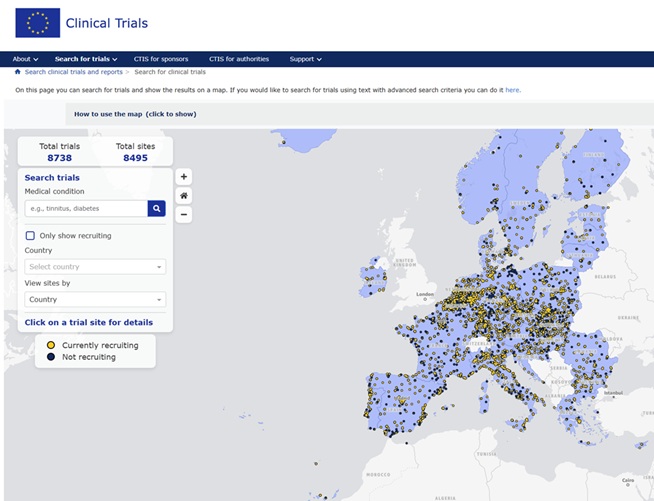
To view the Clinical Trials authorised and active in the EU, in the interactive map created by the Accelerating Clinical Trials in the European Union (ACT EU), please visit the
You can find, in real time, which Clinical Trials are active in the EU and which of them are recruiting patients by searching for the pathologies of interest to you and the country/countries in which they are being conducted. You will be shown the centers where the trials on the pathology you are looking for are being conducted, with their address, clinical trial name and contact details of the main investigator.